
Industries with many regulatory requirements, like life sciences and biosciences companies, have experienced significant changes in the regulatory environment in the past decade. Both implementing this international standard and becoming ISO 9001 certified were shown to increase financial performance. The ISO determined that ISO 9001 certification led to increased sales, which they believe could be due to signaling theory. Customers in these situations look for “signals” of quality, such as certification to a quality standard or management system standards that showcase a commitment to quality. According to signaling theory, which was first articulated in the 1970s, there is an information gap, where customers don’t understand the ins and outs of a product or the processes that go into manufacturing it. There were also benefits that the ISO attributed to the way certification “signaled quality” to potential customers and partners. Better process controls, productivity targets, and improved product quality all helped to increase customer satisfaction and drive sales. Some of these benefits, the ISO reported, were a direct result of the quality initiatives undertaken by these companies to become certified. Additionally, a successful implementation drove external benefits as well, including increased sales and access to new markets. The analysis found that implementation of the ISO 9001 standards delivered internal benefits to lower costs, such as increasing product control, efficiency, and productivity. In 2012, for instance, the International Organization for Standardization (ISO) performed a meta-analysis of published studies to determine whether companies who were certified to ISO 9001 saw any measurable impact on their financial performance. Studies have also shown that a commitment to quality has reputational advantages that can drive sales and increase revenue. This is of paramount importance for biotech and life science companies. ISO 31000 cannot be used for certification, however.Īs discussed earlier, a major benefit of adopting and ISO standard is to ensure that your company consistently delivers a trustworthy experience to your customers. ISO 31000 was developed to set standards within your risk management process for internal auditing and external auditing. Risk management is important for all life science companies.
#Difference between iso 9001 and iso 13485 software#
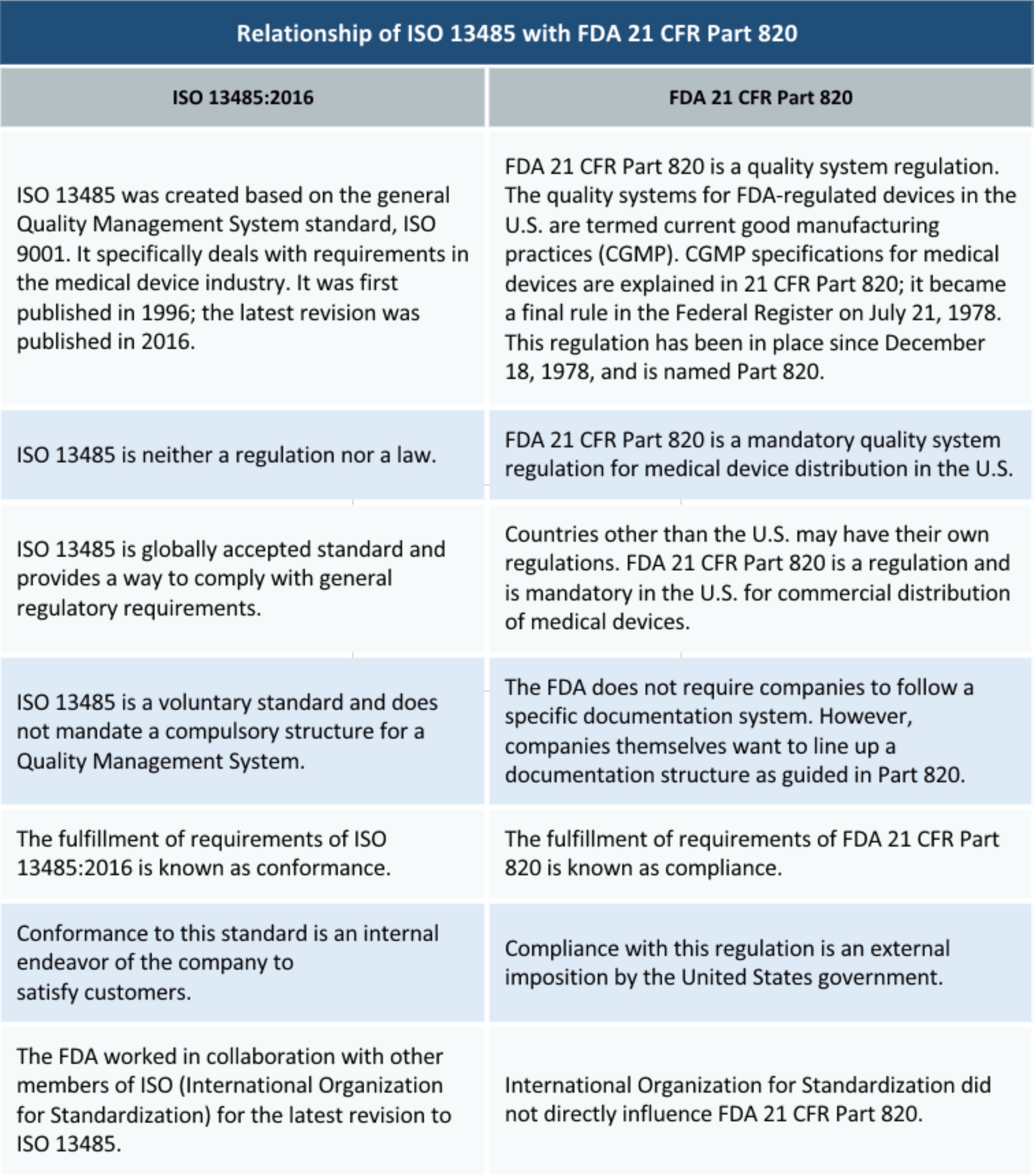
According to ISO, this standard “specifies terminology, principles and a process for risk management of medical devices, including software as a medical device and in vitro diagnostic medical devices.” ISO 31000 - Risk Management ISO 14971 has been widely adopted by the medical device industry and is often a compliance requirement for products with international offerings. ISO 14971 - Risk Management for Medical Devices The ISO 9001 Academy from Advisera has a detailed explanation of the differences between ISO 13485:2016 and ISO 9001:2015. Because ISO 13485 was written specifically for medical device manufacturers (while ISO 9001 is reported to be more generally applied), it has additional requirements to be aware of. The latest release of ISO 13485:2016 was released to support an earlier structure of ISO 9001 before it was updated to ISO 9001:2015. While ISO 9001 and ISO 13485 have similarities, it is important to know that there are also significant differences. If you make or distribute medical devices, you will be best suited to implement ISO 13485. This standard is often considered the first step in meeting EU regulations for medical devices. The ISO 13485:2016 standard is the most important standard for medical device companies to be aware of compared to ISO 9001. ISO 13485:2016 - Quality Management Systems for Medical Devices Let’s take a quick look at these standards.
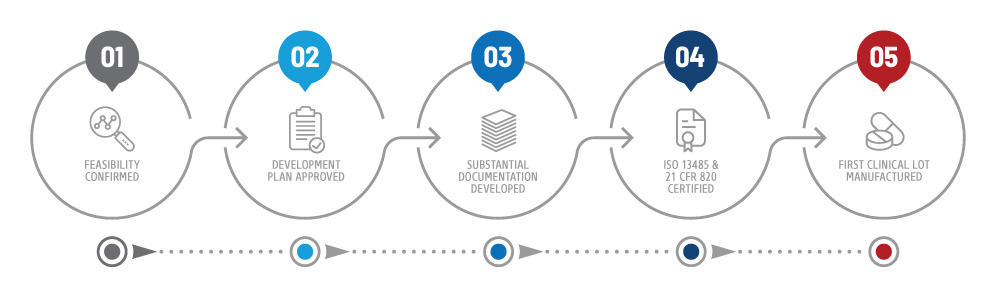
Medical and life science companies may also want to be aware of three other ISO standards: ISO 13485:2016 Medical devices - Quality management systems, ISO 14971:2019 Medical devices - Application of risk management to medical devices ISO 31000.

However, the ISO has published over 20,000 different standards for technology and manufacturing industries ( see ISO's most popular standards), and some of them are of particular importance to medical companies. The ISO 9001 standard is capable of applying to companies in any industry.
ISO 9001 is a set of standards published by the International Organization for Standardization or ISO.
